Evidence of Work
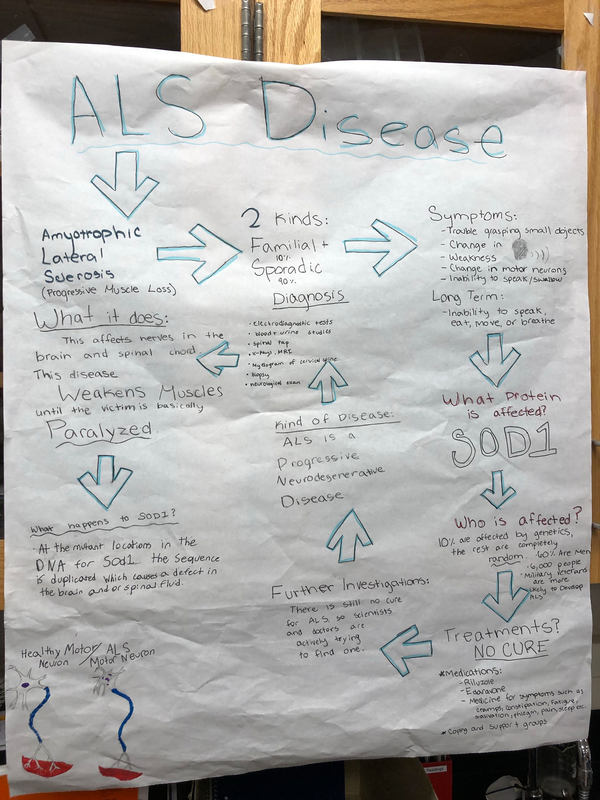
Als (Amyotrophic lateral Sclerosis) is a progressive neurodegenerative disease that affects nerve cells in the brain and the spinal cord. It weakens your muscles and after a while you are basically paralyzed. The symptoms are trouble grasping small objects, change in voice, weakness, change in motor neurons, inability to speak or swallow, and people may not be able to speak, eat, move, or breathe.
Content
Protein Synthesis:
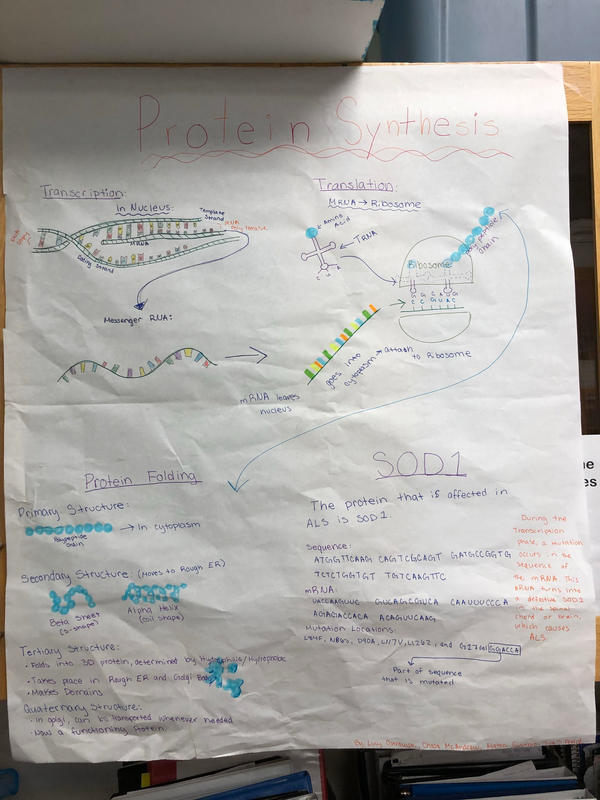
Transcription- in nucleus, mRNA is made and leaves nucleus, then attaches to a ribosome.
Translation- on ribosome, transfer RNA comes to ribosome, bringing an amino acid, amino acids become protein, proteins fold into a specific shape, and make up a functioning protein.
Protein Folding: A protein folds to perform its biological function. There are four stages of protein folding, primary, secondary, tertiary and quaternary. It starts in the cytoplasm. The primary structure is the sequence of amino acids held together by peptide bonds. It then moves into the rough ER. The secondary structure is the protein beginning to fold up. It can have two types of structure: the alpha helix, a coil shape held by hydrogen bonds in the same direction as the coil. The beta sheet is an S shape pattern, also with hydrogen bonds holding the structure together. The hydrogen bonds are between NH and CO groups on the peptides. The tertiary structure is the protein folded into its precise 3D structure, relating to the function. Hydrophilic or hydrophobic dictates more folding. This takes place in the ER and golgi body. This is held together by a range of non-covalent interactions between side groups, including ionic interactions, disulphide bridges, hydrophobic interactions, Van der Waals forces and hydrogen bonds. The quaternary structure is when single peptides bond to other peptides, for example in haemoglobin. This happens in the golgi body.
Vocabulary
Transcription- in nucleus, mRNA is made and leaves nucleus, then attaches to a ribosome.
Translation- on ribosome, transfer RNA comes to ribosome, bringing an amino acid, amino acids become protein, proteins fold into a specific shape, and make up a functioning protein.
Protein Folding: A protein folds to perform its biological function. There are four stages of protein folding, primary, secondary, tertiary and quaternary. It starts in the cytoplasm. The primary structure is the sequence of amino acids held together by peptide bonds. It then moves into the rough ER. The secondary structure is the protein beginning to fold up. It can have two types of structure: the alpha helix, a coil shape held by hydrogen bonds in the same direction as the coil. The beta sheet is an S shape pattern, also with hydrogen bonds holding the structure together. The hydrogen bonds are between NH and CO groups on the peptides. The tertiary structure is the protein folded into its precise 3D structure, relating to the function. Hydrophilic or hydrophobic dictates more folding. This takes place in the ER and golgi body. This is held together by a range of non-covalent interactions between side groups, including ionic interactions, disulphide bridges, hydrophobic interactions, Van der Waals forces and hydrogen bonds. The quaternary structure is when single peptides bond to other peptides, for example in haemoglobin. This happens in the golgi body.
Vocabulary
- Codon/Anti-codon
- Polymerse
- Polypeptide Chain
- Proteins
- 4 proteins folding structures
- Translation
- Transcription
- Alpha Helix
- Beta Sheet
Reflection
This project was actually pretty fun. I really like my group with Mateo and Lucy. I need to work more during the projects instead of letting other people take charge. I did get off topic sometimes and mess with the table group around me. I liked doing the poster because it was easy to get everyone doing something at the same time. Like I would make a picture while Mateo or Lucy was writing.
Overall this was a good project beacuse I feel like we really bonded as group mates.
Overall this was a good project beacuse I feel like we really bonded as group mates.